Blogs

Liquid-Borne Particle Counter Standard Operating Procedure: Principles of Particle Detection, Light Obscuration and Light Scattering Techniques, and Critical Importance in Pharmaceutical Quality Control and Materials Science
A technical standard operating procedure (SOP) reference for liquid-borne particle counters. Explains light obscuration and light scattering principles, measurement techniques, calibration requirements, and applications in pharmaceutical parenteral testing, cleaning validation, and materials science.
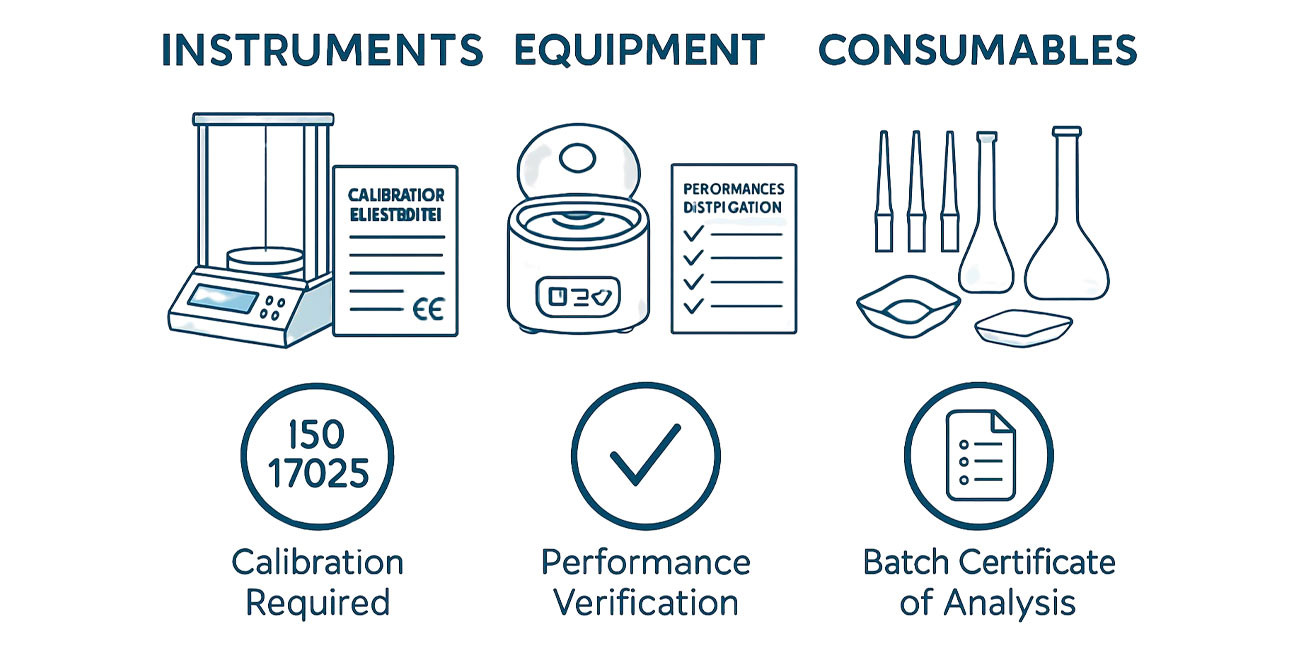
Laboratory Instruments, Equipment, and Consumables: Definitions, Quality Measures, Certification Standards (CE, ISO), and the Critical Role of Precision in Pharmaceutical Analysis
A technical reference article defining laboratory instruments, equipment, and consumables in pharmaceutical analysis. Explains quality measures, precision requirements, CE and ISO certification importance, and whether scientists may use uncertified instruments. Targets analytical chemists and pharmaceutical professionals in Bangladesh.

Laboratory Instrumentation in Modern Scientific Research: Classification, Functions, and Applications of Critical Analytical Instruments
A technically detailed examination of laboratory instrumentation used across scientific research disciplines. This article covers laboratory classification systems, the structural organization of pharmaceutical laboratories, core analytical methods, the operating principles of critical instruments, GMP and DGDA regulatory requirements applicable in Bangladesh, and the outlook for laboratory science in the context of expanding pharmaceutical sector demands.
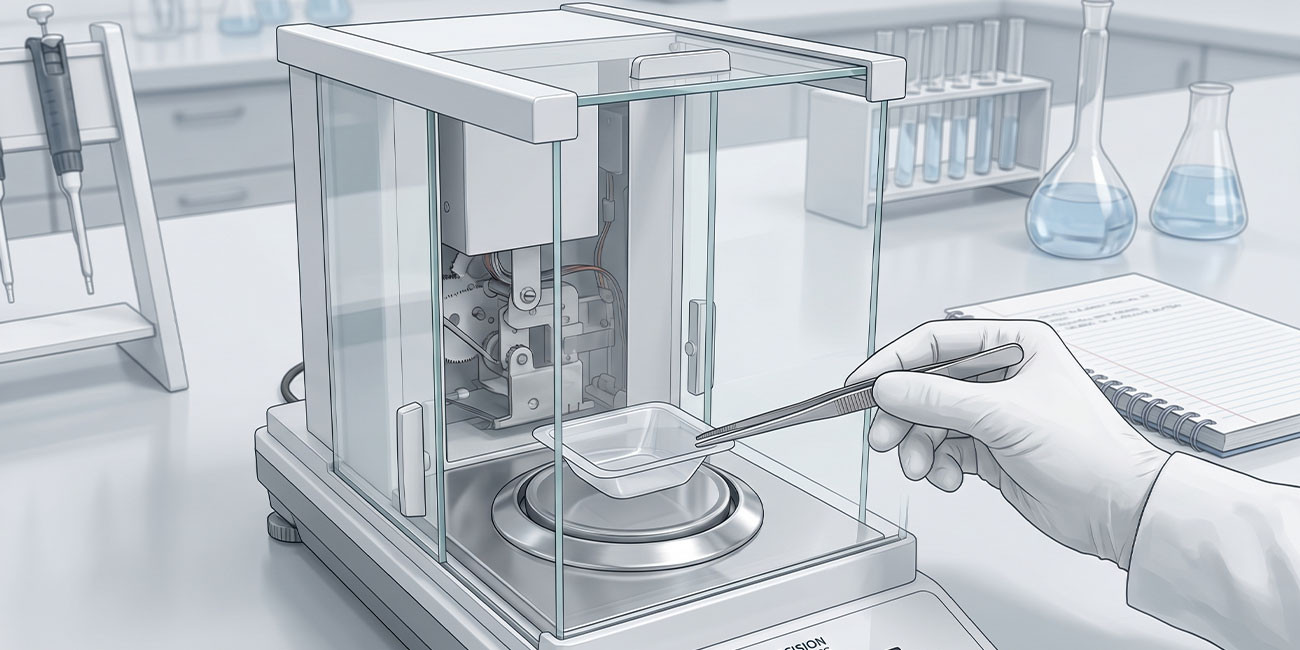
Precision Weighing in Analytical Chemistry: Principles of Laboratory Balances and Measurement Accuracy
A comprehensive technical examination of precision weighing principles in analytical chemistry, focusing on laboratory balance operation, measurement uncertainty, calibration protocols, and regulatory compliance requirements for pharmaceutical laboratories in Bangladesh.

Gas Analysis and Monitoring: Principles of Multi-Gas Detection and Environmental Applications
A technically rigorous examination of gas analysis and multi-gas detection systems used in pharmaceutical laboratories, environmental monitoring programs, and industrial safety applications. This article explores sensor technologies, detection principles, instrument classification, regulatory requirements, and the relevance of gas monitoring infrastructure to pharmaceutical laboratory operations in Bangladesh and international scientific contexts.

Spectroscopic Techniques in Chemical Analysis: Principles, Types, and Laboratory Applications
An in-depth technical guide on spectroscopic techniques and their role in pharmaceutical laboratory Bangladesh, covering analytical methods, instrumentation, regulatory frameworks, and future prospects.

Laboratory Mixing and Shaking Systems: Mechanical Principles and Applications in Scientific Research
Comprehensive analysis of laboratory mixing and shaking systems used in pharmaceutical and analytical chemistry research. This technical guide explores mechanical principles, operational parameters, instrumentation, and regulatory compliance for laboratory agitation equipment in Bangladesh and international pharmaceutical environments.

Pharmaceutical Laboratories in Bangladesh: Analytical Parameters, Testing Methods, and Laboratory Instrumentation
An in-depth examination of pharmaceutical laboratories in Bangladesh, covering their structure, analytical techniques, key instrumentation, GMP compliance under DGDA, and the sector’s critical role in ensuring drug safety, efficacy, and export competitiveness.

Sterility Testing and Contamination Control in Pharmaceutical and Microbiological Laboratories
A deep-dive into sterility testing and contamination control within pharmaceutical QC labs. This article explores regulatory mandates (DGDA, GMP), instrumental methods (HPLC, membrane filtration), facility design (ISO 5/7/8), and the specific challenges and growth trajectory of the pharmaceutical laboratory sector in Bangladesh.

Development of Scientific Laboratory Infrastructure in Bangladesh: Challenges, Standards, and Future Directions
A technical analysis of the evolution of pharmaceutical laboratory infrastructure in Bangladesh, detailing organizational structure, analytical methodologies, and the regulatory landscape.

UV-Visible Spectrophotometry: Instrumentation, Working Principle, and Applications in Chemical Analysis
A technical examination of UV-Visible Spectrophotometry, its working principles, and its vital implementation within the pharmaceutical laboratory infrastructure of Bangladesh.

Analytical Techniques in Pharmaceutical Sciences: Principles, Applications, and Laboratory Implementation
A technical examination of analytical techniques in pharmaceutical sciences, focusing on laboratory structure, instrumentation principles, and regulatory compliance within the Bangladesh pharmaceutical industry.
Recent Posts
Gas Analysis and Monitoring: Principles of Multi-Gas Detection and Environmental Applications
Spectroscopic Techniques in Chemical Analysis: Principles, Types, and Laboratory Applications
Laboratory Mixing and Shaking Systems: Mechanical Principles and Applications in Scientific Research
Sterility Testing and Contamination Control in Pharmaceutical and Microbiological Laboratories
Optimum Solution– Laboratory & Analytical Instruments Supplier in Bangladesh
Optimum Solution is a leading supplier of laboratory equipment, analytical instruments, and process control solutions in Bangladesh. Since our inception, we have built a strong presence in the industry, serving research labs, universities, pharmaceutical companies, and quality control laboratories nationwide. Through continuous dedication to improving our products and services, Optimum Solution has earned the trust and loyalty of our valued clients.
