Laboratory Instruments, Equipment, and Consumables: Definitions, Quality Measures, Certification Standards (CE, ISO), and the Critical Role of Precision in Pharmaceutical Analysis
A laboratory is a controlled environment designed for scientific investigation, measurement, and analysis. In the pharmaceutical context, laboratories generate quantitative data that determine raw material acceptability, process control, and final product quality. The fundamental output of any pharmaceutical laboratory is reliable analytical results that support decisions about batch release, stability, and regulatory compliance.
The purpose extends beyond simple testing. Laboratories establish measurement traceability to international standards, validate analytical methods, investigate out-of-specification results, and maintain documented evidence of quality. Without a properly equipped and managed laboratory, pharmaceutical manufacturing operates without objective quality assurance.
Role in Drug Development and Quality Assurance
During drug development, laboratories characterize active pharmaceutical ingredients, develop stability-indicating methods, and perform forced degradation studies. In quality assurance, laboratories conduct batch release testing, raw material verification, and stability monitoring. The laboratory serves as the final checkpoint before a product reaches patients.
Structure of a Laboratory
Laboratory Sections
Quality Control (QC) Laboratory
Performs routine testing of incoming materials, in-process samples, and finished products. Maintains calibrated instruments and validated methods.
Quality Assurance (QA) Laboratory
Conducts stability studies, method validation verification, and investigative testing. Oversees documentation and audit readiness.
Research and Development (R&D) Laboratory
Develops new analytical methods, performs method transfers, and characterizes reference standards.
Microbiology Laboratory
Conducts sterility testing, microbial limit tests, endotoxin detection, and environmental monitoring under controlled conditions.
Stability Laboratory
Manages accelerated and long-term stability studies under controlled temperature and humidity conditions.
Workflow and Laboratory Design Considerations
Sample flow follows a unidirectional path: receipt, preparation, analysis, review, and disposal. Preparation areas contain fume hoods and balance enclosures. Analytical zones house sensitive instruments on vibration-damped benches. Environmental parameters require active monitoring: temperature 20-25°C, relative humidity 40-60 percent.
Analytical Methods in Pharmaceutical Laboratories
Chemical Analysis Techniques
Titrimetric analysis determines analyte concentration through volumetric reactions. Acid-base, complexometric, redox, and argentometric titrations serve different analyte types. Gravimetric analysis isolates and weighs analytes, providing primary measurement without calibration curves.
Instrumental Methods
Spectroscopic techniques include UV-Visible, Fourier-transform infrared, and atomic absorption spectroscopy. Chromatographic methods include high-performance liquid chromatography, gas chromatography, and ion chromatography. Electrochemical methods include potentiometry and Karl Fischer titration.
Sample Preparation and Validation
Sample preparation converts the test portion into a solution suitable for analysis. Steps include dissolution, extraction, dilution, and filtration. Method validation demonstrates accuracy, precision, specificity, detection limit, quantitation limit, linearity, range, and robustness.
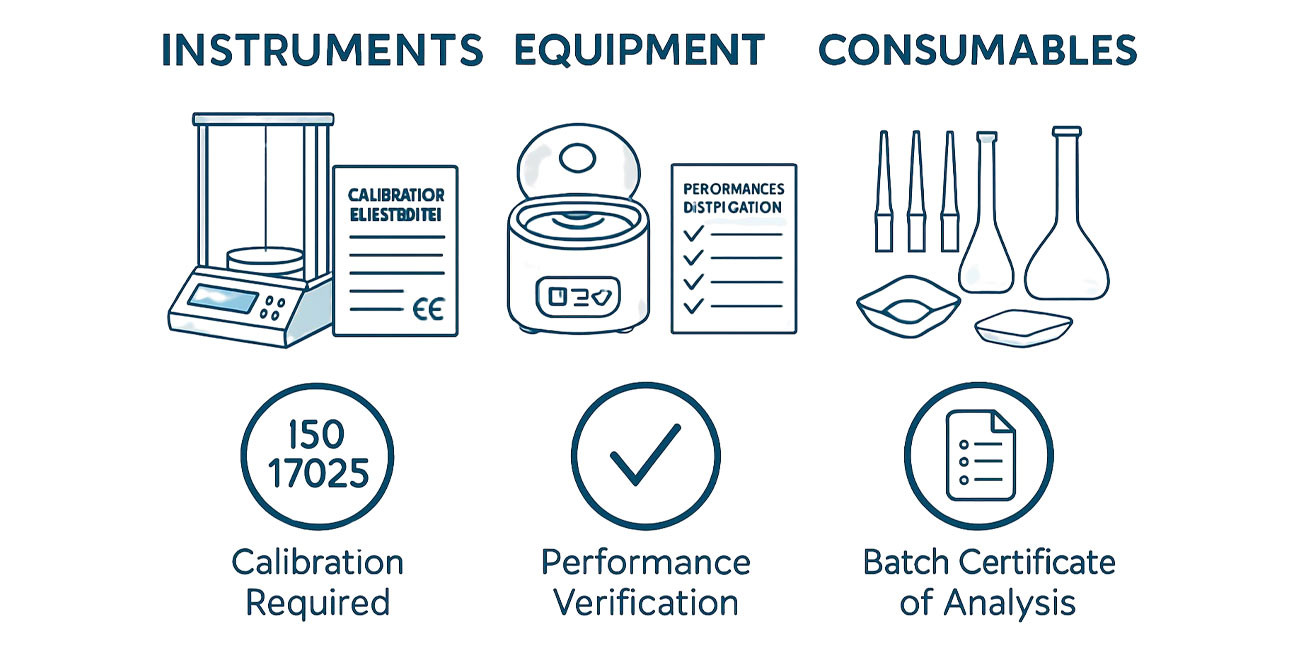
Laboratory Instrumentation – Definitions and Classifications
Definition of Laboratory Instruments
Laboratory instruments are precision devices that measure physical or chemical properties of a sample and convert those properties into quantifiable signals. Instruments require calibration, verification, and maintenance to produce valid data. Examples include analytical balances, pH meters, spectrophotometers, and chromatographs.
Key characteristics of instruments:
Contain electronic or optical components
Require regular calibration against reference standards
Produce a measurable output (mass, absorbance, potential, retention time)
Operate according to defined physical principles (electromagnetic force compensation, Beer-Lambert law, Nernst equation)
Performance verification required before each use or on a scheduled basis
Definition of Laboratory Equipment
Laboratory equipment refers to apparatus that supports sample handling, preparation, or environmental control but does not directly measure an analyte. Equipment may require verification of proper operation but typically does not produce quantitative measurements. Examples include fume hoods, centrifuges, water baths, ovens, incubators, and refrigerators.
Distinguishing features of equipment:
Support or prepare samples rather than measure them
Require performance verification (temperature uniformity, rotational speed accuracy)
Less frequent calibration compared to instruments
Failure may affect sample integrity but not direct measurement
Definition of Laboratory Consumables
Laboratory consumables are single-use or limited-use items that are discarded after one or several applications. Consumables must be free from contaminants that could interfere with analysis. Examples include volumetric flasks, pipette tips, cuvettes, filters, weighing boats, chromatography vials, and disposable gloves.
Critical properties of consumables:
Batch-to-batch consistency required
Certificates of analysis available for critical items (filters, vials)
Proper storage conditions to prevent degradation
Expiration dates for items with limited shelf life (standards, reagents, some plastics)
Distinction Table
Instrument Quality Measures – Standards for a Good Laboratory
What Quality Should Be Maintained in a Good Laboratory
A good laboratory maintains quality across five domains: personnel competence, instrument performance, method validity, environmental control, and documentation integrity.
Personnel Competence
Analysts must demonstrate proficiency through initial training, periodic assessment, and continuing education. Written procedures describe each analytical operation. Supervisors verify analyst performance through split-sample testing and blind replicates.
Instrument Performance
Each instrument requires a documented calibration schedule. Calibration traceable to national or international standards. Performance verification before each use for critical instruments (balance daily check, HPLC system suitability). Out-of-tolerance results trigger investigation and sample reanalysis.
Method Validity
Analytical methods must be validated for their intended use. Parameters include accuracy, precision, specificity, detection limit, quantitation limit, linearity, range, and robustness. Validated methods cannot be modified without revalidation.
Environmental Control
Temperature, humidity, vibration, and air quality monitored and recorded. Sensitive instruments located away from heat sources, air vents, and foot traffic. Balances placed on vibration-damped tables. HPLC solvents filtered and degassed.
Documentation Integrity
All raw data recorded directly, dated, and signed. Electronic records maintain audit trails. Changes documented without obscuring original entries. Retention periods comply with regulatory requirements.
Why Precision is Important
Precision measures the agreement between repeated measurements of the same sample under identical conditions. In analytical chemistry, precision is expressed as standard deviation, relative standard deviation, or variance.
Mathematical Definition
For a set of n measurements (x₁, x₂, ..., xₙ) with mean x̄:
Standard deviation s = √[Σ(xᵢ - x̄)²/(n-1)]
Relative standard deviation RSD = (s/x̄) × 100%
Impact on Measurement Uncertainty
Precision directly contributes to total measurement uncertainty. Poor precision widens confidence intervals. For a 95% confidence interval, the half-width equals t × s/√n, where t is Student's t-value. A two-fold increase in standard deviation doubles the confidence interval width.
Consequences of Poor Precision
In assay determination, RSD exceeding 2% may produce batch failure or false acceptance. In content uniformity testing, poor precision masks actual dosage form variation. In impurity analysis, low precision near the quantitation limit leads to false positives or negatives.
Precision Requirements by Method Type
Analytical balance weighing: RSD ≤ 0.1% for 1 mg loads
HPLC assay: RSD ≤ 1.0% for six injections
UV-Vis absorbance: RSD ≤ 0.5% for replicate readings
Titration: RSD ≤ 0.2% for volume measurements
Why Laboratory Items Should Be CE or ISO Certified
CE Marking (Conformité Européenne)
CE marking indicates that a product meets European Union safety, health, and environmental protection requirements. For laboratory instruments, CE certification covers electrical safety, electromagnetic compatibility, and mechanical hazards.
A CE-marked balance includes protection against electrostatic discharge, radiated emissions, and electrical fast transients. A non-CE instrument may generate electromagnetic interference affecting nearby sensitive equipment. CE certification provides documented evidence that the manufacturer has performed hazard analysis and risk assessment.
ISO Certification for Instruments
ISO 9001 certifies that the manufacturer operates a quality management system. For laboratory instruments, ISO 9001 ensures consistent production quality, documented design controls, and corrective action processes.
ISO/IEC 17025 applies specifically to testing and calibration laboratories. Instruments manufactured under ISO 17025 accreditation come with calibration certificates traceable to international standards.
Why Certification Matters
Traceability: Certified instruments provide documented traceability to national metrology institutes
Regulatory acceptance: GMP inspectors expect ISO-certified instruments or documented equivalency
Inter-laboratory comparability: ISO-compliant instruments produce results comparable across laboratories
Risk reduction: Certification reduces probability of manufacturing defects, calibration drift, or safety hazards
Specific Requirements for Pharmaceutical Laboratories in Bangladesh
DGDA GMP guidelines reference ISO standards. WHO prequalification requires instruments meeting international safety and performance standards. Export to EU markets requires CE-marked instruments for regulatory submission acceptance.
Should Any Scientist Use Instruments Without Certification?
Direct Answer: No, with limited exceptions.
Reasons Against Using Uncertified Instruments
Data integrity risk: An uncertified instrument produces measurements without established traceability. If challenged during inspection, the laboratory cannot prove the instrument met performance specifications at the time of testing.
Regulatory violation: GMP clauses require calibrated instruments with documented traceability. Using uncertified instruments constitutes non-compliance. Inspection findings may include 483 observations or warning letters.
Patient safety impact: Uncertified balances produce inaccurate sample weights. A 1% error in potent drug weighing could cause 10% concentration error in final product if compounded through subsequent dilutions.
Legal liability: In case of product failure leading to patient harm, use of uncertified instruments constitutes negligence. Legal discovery would reveal the absence of certification.
Limited Exceptions
Research and development (non-GMP): In early-stage R&D where results do not support regulatory submissions, uncertified instruments may be used for screening or qualitative work. However, any data entering a regulatory filing must come from certified instruments.
Educational settings: Teaching laboratories may use uncertified instruments for demonstration purposes, provided students learn the distinction between educational and regulatory environments.
Validation against certified reference: An uncertified instrument may be used temporarily if its performance is verified daily against certified reference standards and results are not used for batch release. This applies only until a certified instrument becomes available.
Risk-Based Approach
The decision matrix for using uncertified instruments:
Corrective Action
If a laboratory currently uses uncertified instruments for GMP testing, immediate actions include:
Stop using the instrument for regulatory testing
Assess risk to previously tested batches
Requalify or replace the instrument with certified equipment
Implement instrument qualification protocol (IQ/OQ/PQ) for all future purchases
Regulatory Framework in Bangladesh
Overview of Regulatory Bodies
Directorate General of Drug Administration (DGDA)
Issues manufacturing licenses, conducts GMP inspections, and evaluates laboratory compliance. DGDA inspectors verify instrument calibration records, certification documents, and performance verification logs.
Bangladesh Standards and Testing Institution (BSTI)
Provides calibration services and sets standards for laboratory equipment.
GMP Requirements for Laboratory Instruments
Schedule M (revised) of the Drug Rules 1945 specifies instrument requirements:
Balances: Appropriate range and precision, calibrated daily with certified weights
pH meters: Calibrated before each use with two buffers
Spectrophotometers: Wavelength accuracy verified quarterly
Chromatographs: System suitability before each sequence
Instruments without valid calibration certificates cannot be used for GMP testing.
International Standards
WHO GMP requires instruments meeting international specifications. USP <1058> describes analytical instrument qualification (AIQ) phases: Design Qualification, Installation Qualification, Operational Qualification, Performance Qualification. ISO/IEC 17025 requires instruments calibrated against measurement standards traceable to SI units.
Importance of Pharmaceutical Laboratories in Bangladesh
Drug Safety and Efficacy
Laboratory testing prevents substandard products from reaching patients. Accurate instruments detect impurities, verify potency, and confirm dissolution profiles. Certification of instruments ensures measurement reliability across all testing.
Industrial Growth
Bangladesh pharmaceutical industry exports to over 150 countries. Export registration requires demonstrated laboratory competence, including certified instruments. Buyer audits specifically examine instrument calibration and certification status.
Export Quality Assurance
Laboratories supporting export operations maintain ISO/IEC 17025 accreditation. Instruments used for export batch testing must have CE marking or equivalent certification (FDA listing, Canadian registration). Non-certified instruments automatically disqualify an export application.
Public Health Impact
Certified instruments produce reliable data for market surveillance. The National Drug Testing Laboratory uses ISO-certified instruments for government sample analysis. Public confidence depends on laboratory measurement integrity.
Challenges and Future Outlook
Current Challenges in Bangladesh
Certified instrument availability: Limited local suppliers for CE-marked and ISO-certified instruments
Calibration infrastructure: National calibration laboratories cannot accommodate all instrument types
Cost constraints: Certified instruments cost 30-50% more than uncertified alternatives
Awareness gaps: Some laboratory managers do not distinguish between equipment and instruments
Consumable quality: Locally sourced consumables often lack certificates of analysis
Technological Advancements
Self-diagnostic instruments: Balances with internal calibration and performance verification
Blockchain traceability: Digital certificates for instrument calibration history
Cloud-based calibration management: Automated scheduling and documentation
Future Opportunities
Local certification bodies: Development of Bangladesh-based ISO 17025 calibration laboratories
Regional harmonization: SAARC mutual recognition of instrument certification
Training programs: University curricula incorporating instrument qualification principles
Recent Posts
Gas Analysis and Monitoring: Principles of Multi-Gas Detection and Environmental Applications
Spectroscopic Techniques in Chemical Analysis: Principles, Types, and Laboratory Applications
Laboratory Mixing and Shaking Systems: Mechanical Principles and Applications in Scientific Research
Sterility Testing and Contamination Control in Pharmaceutical and Microbiological Laboratories
OS4U – Laboratory & Analytical Instruments Supplier in Bangladesh
OS4U is a leading supplier of laboratory equipment, analytical instruments, and process control solutions in Bangladesh. Since our inception, we have built a strong presence in the industry, serving research labs, universities, pharmaceutical companies, and quality control laboratories nationwide. Through continuous dedication to improving our products and services, OS4U has earned the trust and loyalty of our valued clients.