Precision Weighing in Analytical Chemistry: Principles of Laboratory Balances and Measurement Accuracy
Pharmaceutical laboratories serve as the analytical backbone of drug development, manufacturing, and quality assurance operations. These specialized facilities generate quantitative and qualitative data that determine whether raw materials, intermediates, and finished pharmaceutical products meet predefined specifications.
The fundamental purpose of a pharmaceutical laboratory is to establish objective evidence of product quality, safety, and efficacy through systematic analytical testing. Without reliable laboratory data, pharmaceutical manufacturing operates without quality control, introducing unacceptable risks to patient health.
Role in Drug Development and Quality Assurance
In drug development, pharmaceutical laboratories conduct method development, validation studies, and stability testing to characterize drug substances and formulations. During commercial manufacturing, quality control laboratories perform batch release testing, raw material verification, and in-process control analyses.
Quality assurance laboratories monitor environmental conditions, validate cleaning procedures, and investigate out-of-specification results. The integration of laboratory data into manufacturing decisions directly impacts product release, batch disposition, and corrective action implementation.
Structure of a Pharmaceutical Laboratory
A pharmaceutical laboratory operates as a system of specialized sections, each designed for specific analytical functions with defined workflows and environmental controls.
Laboratory Sections
Quality Control (QC) Laboratory
Performs routine testing of raw materials, packaging components, in-process samples, and finished products. Maintains validated analytical methods and calibrated instruments.
Quality Assurance (QA) Laboratory
Conducts stability studies, method validation verification, and investigative testing. Oversees documentation systems and audit readiness.
Research and Development (R&D) Laboratory
Develops new analytical methods, performs forced degradation studies, and characterizes reference standards. Supports formulation development through analytical testing.
Microbiology Laboratory
Conducts sterility testing, microbial limit tests, endotoxin detection, and environmental monitoring. Operates under controlled contamination prevention protocols.
Stability Laboratory
Manages accelerated and long-term stability studies under controlled temperature and humidity conditions. Performs periodic testing according to stability protocols.
Workflow and Laboratory Design Considerations
Sample workflow follows a unidirectional path from receipt to disposal to prevent cross-contamination. Receiving areas log samples into laboratory information management systems. Preparation areas contain fume hoods and balance enclosures. Analytical zones house sensitive instruments on vibration-damped benches. Washing and waste disposal areas operate separately from analytical spaces.
Environmental parameters require active monitoring. Temperature maintained at 20-25°C for general laboratories, 2-8°C for cold storage. Relative humidity controlled at 40-60 percent to prevent hygroscopic sample issues. Vibration sources isolated from precision balances and chromatographs.
Analytical Methods in Pharmaceutical Laboratories
Analytical methods translate chemical and physical properties into measurable signals that correlate with sample composition. Each method requires validation to demonstrate suitability for its intended purpose.
Chemical Analysis Techniques
Titrimetric analysis determines analyte concentration through volumetric reactions. Acid-base titrations quantify basic or acidic functional groups. Complexometric titrations using EDTA determine metal ion concentrations. Redox titrations measure oxidizing or reducing agents. Argentometric methods quantify halides through silver nitrate reactions.
Gravimetric analysis isolates and weighs analytes after precipitation, volatilization, or extraction. This primary method provides direct mass measurement without calibration curves, serving as reference technique for method validation.
Instrumental Methods
Spectroscopic Techniques
Ultraviolet-visible spectroscopy measures light absorption at specific wavelengths to quantify chromophoric compounds. Fourier-transform infrared spectroscopy identifies functional groups through molecular vibration patterns. Atomic absorption spectroscopy determines elemental concentrations at parts-per-million levels.
Chromatographic Techniques
High-performance liquid chromatography separates, identifies, and quantifies components in complex mixtures. Reverse-phase HPLC dominates pharmaceutical analysis due to compatibility with drug substances. Gas chromatography analyzes volatile compounds with thermal stability. Ion chromatography measures inorganic ions and organic acids.
Electrochemical Methods
Potentiometry uses ion-selective electrodes to measure specific ion activities. pH measurement represents the most common potentiometric application. Karl Fischer titration specifically determines water content through coulometric or volumetric titration.
Sample Preparation and Validation
Sample preparation converts the test portion into a solution suitable for instrumental analysis. Weighing accuracy directly influences final results. Preparation steps include dissolution, extraction, derivatization, dilution, and filtration. Each step introduces potential error sources requiring control.
Method validation demonstrates analytical procedure suitability through specific parameters:
Accuracy: closeness of measured value to true value
Precision: agreement between repeated measurements
Specificity: ability to measure target analyte without interference
Detection limit: lowest analyte quantity that can be detected
Quantitation limit: lowest analyte quantity that can be measured with precision
Linearity: proportional relationship between signal and concentration
Range: interval between upper and lower concentrations with acceptable accuracy and precision
Robustness: method reliability under small variations in conditions
Laboratory Instrumentation
Pharmaceutical laboratories require precisely specified instruments performing defined functions. Each instrument operates on established scientific principles with documented calibration and maintenance requirements.
Key Instruments and Functions
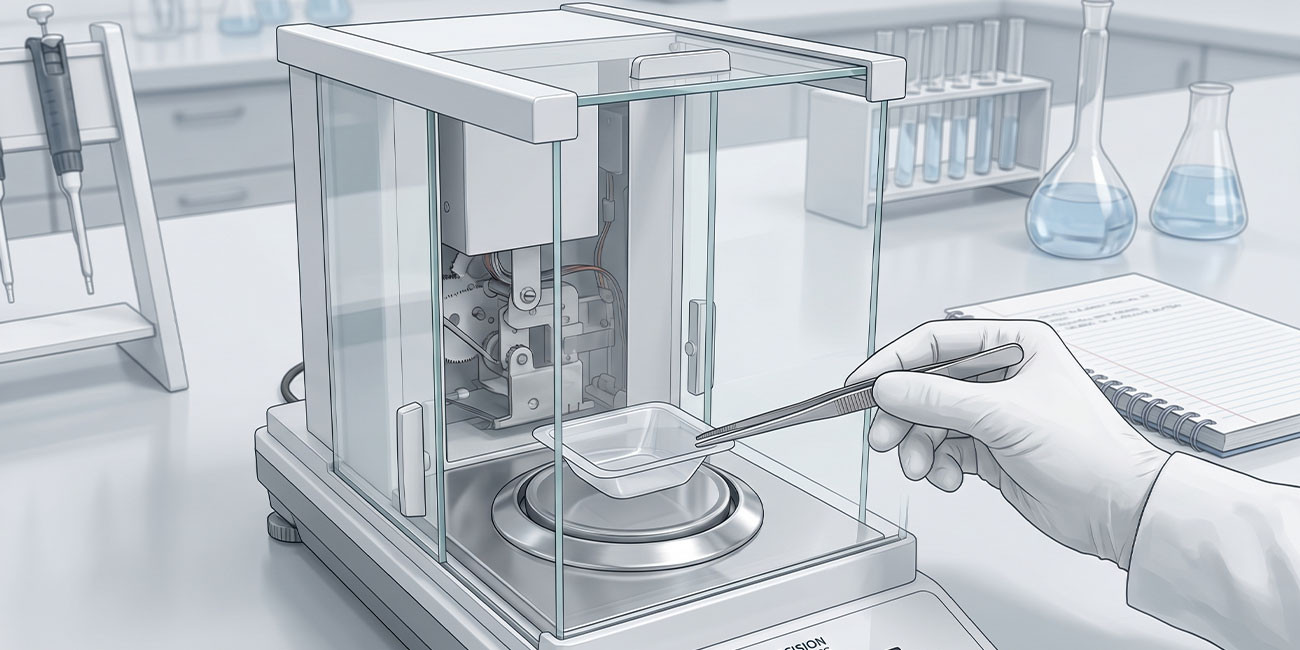
Analytical Balances
Readability of 0.1 mg or 0.01 mg for analytical work. Internal calibration mechanisms adjust for temperature and gravity variations. Draft shields prevent air currents from affecting measurements.
pH Meters
Measure hydrogen ion activity with resolution of 0.001 pH units. Temperature compensation adjusts for Nernst equation temperature dependence. Calibration requires minimum two buffer solutions bracketing sample pH.
UV-Visible Spectrophotometers
Measure absorbance from 190 to 1100 nm. Deuterium lamps provide ultraviolet radiation. Tungsten lamps provide visible radiation. Double-beam designs compensate for source intensity variations.
HPLC Systems
Solvent delivery pumps maintain flow rates from 0.1 to 10 mL/min at pressures to 6000 psi. Autosamplers inject precise volumes. Column ovens control temperature within 0.1°C. Detectors include UV-visible, diode array, fluorescence, and evaporative light scattering.
Dissolution Test Apparatus
Paddle and basket methods simulate drug release in gastrointestinal fluids. Sampling at specified intervals generates dissolution profiles. Automated systems collect samples at programmed time points.
Principle of Operation for Each Instrument
Analytical balances operate on electromagnetic force compensation principles. A coil carrying current generates magnetic force opposing the load. Feedback circuits adjust current to restore balance position. The required current directly proportional to mass.
pH meters measure potential difference between glass indicating electrode and reference electrode. Glass membrane develops potential proportional to hydrogen ion activity difference between internal buffer and sample.
HPLC separations occur through differential partitioning between mobile phase and stationary phase. Pump forces mobile phase through packed column. Analytes interact with stationary phase based on polarity, producing retention time differences.
Importance in Pharmaceutical Testing
Instrumentation provides quantitative data supporting regulatory submissions and batch release decisions. HPLC quantifies active pharmaceutical ingredient content and related substances. Balances ensure accurate sample weights for all subsequent analyses. Dissolution testers measure drug release characteristics affecting bioavailability.
Instrument performance directly impacts patient safety. Incorrect potency measurements could lead to underdosing or overdosing. Undetected impurities could cause adverse reactions. Reliable instrumentation with documented performance verification protects public health.
Regulatory Framework in Bangladesh
Pharmaceutical laboratories in Bangladesh operate within a regulatory system that establishes standards, conducts inspections, and enforces compliance requirements.
Overview of Regulatory Bodies
Directorate General of Drug Administration (DGDA)
Primary drug regulatory authority under Ministry of Health and Family Welfare. Issues manufacturing licenses, product registration certificates, and import permits. Conducts Good Manufacturing Practice inspections of pharmaceutical facilities. Evaluates laboratory compliance through audit and sample testing.
Bangladesh Standards and Testing Institution (BSTI)
Sets standards for pharmaceutical packaging and raw materials. Provides calibration services for laboratory equipment.
Good Manufacturing Practice Requirements for Laboratories
GMP guidelines specify laboratory requirements in Part I, Chapter 6 of the Bangladesh National Drug Policy. Quality Control laboratories must be physically separated from production areas. Written procedures required for sampling, testing, and record-keeping.
Laboratory GMP requirements include:
Personnel
Qualified analysts with documented training. Job descriptions specifying responsibilities. Continuing education programs for method updates.
Premises and Equipment
Dedicated areas for sensitive instruments. Environmental monitoring programs. Calibration schedules traceable to national standards.
Documentation
Standard operating procedures for all tests. Specification documents for materials and products. Analytical worksheet records for each sample. Investigation reports for out-of-specification results.
Reference Standards
Primary reference standards from pharmacopoeias. Working standards qualified against primary standards. Documentation of standard preparation and storage.
Testing
Validated methods for all parameters. System suitability tests before sample analysis. Replicate determinations for critical tests.
International Standards
World Health Organization (WHO)
WHO Good Manufacturing Practices apply to all pharmaceutical manufacturing in Bangladesh. WHO Prequalification program assesses laboratories for quality and competence.
United States Pharmacopoeia (USP)
USP-NF provides analytical methods and acceptance criteria. General chapters describe balance requirements, chromatographic system suitability, and analytical method validation.
International Organization for Standardization (ISO)
ISO/IEC 17025 specifies laboratory competence requirements. ISO 9001 addresses quality management systems. ISO 15189 applies to medical laboratories.
Importance of Pharmaceutical Laboratories in Bangladesh
Pharmaceutical laboratories function as quality gatekeepers for the domestic drug supply and export products reaching international markets.
Drug Safety and Efficacy
Laboratory testing prevents substandard and falsified medicines from reaching patients. Identity testing confirms correct raw materials. Assay testing verifies correct potency. Dissolution testing ensures drug release for absorption. Stability testing establishes shelf life and storage conditions.
The National Drug Policy 1982 (revised 2005) mandates testing of all imported raw materials and finished products. Local manufacturers must test each batch before release. Market surveillance samples undergo confirmatory testing at government laboratories.
Industrial Growth
Bangladesh pharmaceutical industry has grown from 10 manufacturers in 1982 to approximately 250 active companies. Domestic market value exceeds 30,000 crore BDT annually. Export earnings reached 1,300 crore BDT in 2022-2023.
Laboratory capability enabled this growth. WHO GMP certification required accredited laboratories. Export registration with regulatory authorities like Therapeutic Goods Administration (Australia) and Medicines and Healthcare products Regulatory Agency (UK) required demonstrated laboratory competence.
Export Quality Assurance
Laboratories supporting export operations maintain standards meeting destination country requirements. Stability studies conducted at International Conference on Harmonisation (ICH) conditions. Impurity profiles documented according to ICH guidelines. Analytical method validation packages submitted with registration dossiers.
Export to regulated markets requires demonstration of analytical equivalence to reference products. Comparative dissolution testing proves similar release characteristics. Forced degradation studies demonstrate stability-indicating method capability.
Public Health Impact
Reliable laboratory testing directly protects patient populations. Detection of impurities like diethylene glycol in glycerin prevented mass poisonings. Potency verification ensures therapeutic effect. Sterility testing prevents contaminated injectable products from reaching hospitals.
The National Drug Testing Laboratory analyzes samples from government hospitals and clinics. Border inspection points screen imported medicines. Quality monitoring programs sample retail pharmacy products.
Challenges and Future Outlook
Pharmaceutical laboratories in Bangladesh face operational and technical challenges while technological advances create opportunities for improvement.
Current Challenges in Bangladesh
Instrumentation Access
High-precision instruments require foreign currency for procurement. Import duties and taxes increase equipment costs. Spare parts availability faces supply chain delays. Service contracts require overseas technical support.
Calibration Infrastructure
National calibration laboratories lack capacity for all instrument types. Many laboratories send balances to Singapore or India for calibration. In-house calibration requires reference weights traceable to certified standards.
Skilled Personnel Shortage
Analytical chemistry programs produce fewer graduates than industry demand. Specialized skills in chromatographic method development remain scarce. English language proficiency limits international regulatory interaction.
Documentation Practices
Manual record-keeping persists in many laboratories. Data integrity issues arise from paper-based systems. Audit trail requirements for electronic records remain poorly understood.
Environmental Control
Temperature fluctuations affect sensitive instruments. Humidity control requires continuous air conditioning operation. Dust levels exceed specifications for balance rooms.
Technological Advancements
Automated Sample Preparation
Robotic systems weigh, dissolve, dilute, and filter samples. Automation reduces analyst variability and improves precision. Twenty-four hour operation increases throughput.
Chromatography Data Systems
Networked systems manage instrument control, data acquisition, and reporting. Audit trails document all user actions. Electronic signatures replace paper approvals.
Real-Time Release Testing
Process analytical technology measures quality attributes during manufacturing. Near-infrared spectroscopy predicts assay and content uniformity. Real-time testing reduces laboratory workload.
Laboratory Information Management Systems
Database systems track samples, results, and stability studies. Automated calculations eliminate manual errors. Dashboard displays provide management visibility.
Future Opportunities in Pharmaceutical Analysis
High-Resolution Mass Spectrometry
Time-of-flight and Orbitrap instruments provide accurate mass measurement. Unknown impurity identification without reference standards. Metabolite profiling for drug development.
Two-Dimensional Chromatography
Heart-cutting and comprehensive techniques separate complex mixtures. Improved resolution for closely related impurities. Reduced analysis time compared to serial methods.
Green Analytical Chemistry
Method development minimizing solvent consumption. Supercritical fluid chromatography reduces organic solvent use. Miniaturized techniques lower sample and reagent requirements.
Regional Laboratory Networks
Shared access to specialized instruments across manufacturers. Proficiency testing programs for method comparison. Joint training initiatives for skill development.
Recent Posts
Gas Analysis and Monitoring: Principles of Multi-Gas Detection and Environmental Applications
Spectroscopic Techniques in Chemical Analysis: Principles, Types, and Laboratory Applications
Laboratory Mixing and Shaking Systems: Mechanical Principles and Applications in Scientific Research
Sterility Testing and Contamination Control in Pharmaceutical and Microbiological Laboratories
Optimum Solution (OS4U) – Laboratory & Analytical Instruments Supplier in Bangladesh
Optimum Solution (OS4U) is a leading supplier of laboratory equipment, analytical instruments, and process control solutions in Bangladesh. Since our inception, we have built a strong presence in the industry, serving research labs, universities, pharmaceutical companies, and quality control laboratories nationwide. Through continuous dedication to improving our products and services, Optimum Solution has earned the trust and loyalty of our valued clients.